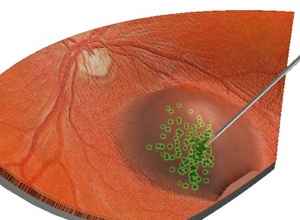
Gene Therapy For Blind Again A Success, More Patients To Be Treated

Share
In 2009, 12 people participated in a clinical trial through which they received an experimental gene therapy for an inherited form of blindness. The correct form of the gene, meant to replace their faulty ones, was injected into the retinal cells of one eye. Limiting the injection to one eye was precautionary, since the researchers didn’t know if there would be any adverse affects. When the trial began all of the participants were legally blind, but two weeks after the injection their vision began to improve. Although their vision didn’t recover enough to be considered normal vision, half of the participants improved enough to lose their legally blind status. Doctors recently revisited the group and have injected the gene into the untreated eyes of three of the original participants. The readministration yielded even further improvement. All three of the participants showed improved vision in dim light as a result, and two of them could even navigate an obstacle course under low-light conditions. Importantly, none of them showed adverse effects to the additional gene injection.
At six months after readministration, the results are so promising that the research team, led by Jean Bennett, an opthalmology professor at the University of Pennsylvania, has begun injecting the gene into the untreated eyes of the remaining participants. The study was published recently in Science Translational Medicine.
Leber congenital amaurosis is caused by a mutation in the gene RPE65. RPE65 normally encodes an enzyme that is produced by cells of the retinal pigment epithelium, the thin layer of cells that supports the health of the retina. In Leber congenital amaurosis, a mutated enzyme fails to convert vitamin A into a form needed for the photosensitive cells of the retina to detect light. The unconverted form of vitamin A builds up and kills cells in the retinal pigment epithelium. Vision loss occurs early, usually within the first six months of a child’s life. And it’s severe. People with Leber congenital amaurosis rarely retain a visual acuity better than 20/400 and far exceed the minimum detriment of 20/200 to be considered legally blind.
Four of the participants were 11 years old or younger at the time of the original trial. Notably, the most significant improvements occurred in the young participants. Researchers speculated that their young retinas might not have gone through the same amount of dystrophy as those of the older participants. This is important in light of the fact that many children with LCA will rub their eyes which can lead to secondary problems.
To further quantify improvement to vision, functional magnetic resonance imaging (fMRI) brain scans were performed while a dim, flickering checkerboard pattern was projected in front of a participant’s recently treated eye. Comparing these scans to scans taken before injection of the second gene they confirmed that the vision-processing areas of the brain were being activated. What’s more, the area of the brain activated by stimulating the first eye showed even greater activity when both eyes were stimulated. The scientists hypothesize that signals now being sent by the second eye were combining with and enhancing signals from the first eye. Both eyes were working together.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


Surprised by this result, the team was relieved by another. It remained possible that the initial injection could have acted like a vaccine, priming the immune system against the second injection. Being “immune-privileged,” however, the eye is more likely to tolerate a foreign substance without eliciting an immune response. Fortunate for the participants, the second injection indeed did not trigger an immune response.
A rare condition, Leber congenital amaurosis affects just 1 person in 80,000. Looking ahead, Bennett hopes to apply the same approach to other more common eye diseases. As for treating Leber congenital amaurosis, they plan to begin enrolling more participants of even younger ages, possibly as young as six months old. If the procedure proves safe in the very young it would be the ideal strategy to treat the disease early, before permanent damage is done to the retina.
The results of the current study are promising not only for the visually impaired but for gene therapies in general, constantly beleaguered by setbacks. Whether or not RPE65’s success can be applied to other types of gene therapies, we can only wait and see.
[image credits: Young Doctors Research Forum, Blindness.org, Nuffield Laboratory of Opthalmology]
image 1: eye test
image 2: Bennett
image 3: retina
Peter Murray was born in Boston in 1973. He earned a PhD in neuroscience at the University of Maryland, Baltimore studying gene expression in the neocortex. Following his dissertation work he spent three years as a post-doctoral fellow at the same university studying brain mechanisms of pain and motor control. He completed a collection of short stories in 2010 and has been writing for Singularity Hub since March 2011.
Related Articles
Brain Implants Let Paralyzed People Type With Thought Alone—Nearly as Fast as Smartphone Users
Digital Twin of a Cell Tracks Its Entire Life Cycle Down to the Nanoscale

This Week’s Awesome Tech Stories From Around the Web (Through March 14)
What we’re reading