Implantable Eye Telescope That Treats AMD Now FDA Approved
Share
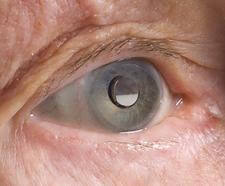
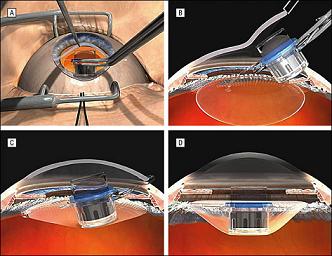
A telescopic implant that fits directly into the eye to treat certain kinds of blindness has finally received FDA approval for use in the US after more than five years of waiting. The Implantable Miniature Telescope (IMT) is used to treat age-related macular degeneration (AMD), a condition that affects millions around the world. For many, the center part of their vision becomes blurred or completely dark. The IMT is surgically implanted into the cornea and acts to expand an incoming image onto the peripheral parts of the retina that are undamaged by AMD. More than two-thirds of those treated enjoy significantly improved vision. The commercial version of the IMT is called CentraSight and is in development by VisionCare Inc. There are likely hundreds of thousands of potential patients in the US alone who may be able to have their vision partially restored now that CentraSight has garnered FDA approval.
Science has no lack of possible cures for blindness. We've seen success with both artificial retinas and stem cell treatments. Each of these 'cures' however, is really just a solution for a very specific kind of vision impairment (the stem cell therapies work best on corneal burns, the artificial retina on certain forms of retinal damage). The CentraSight implant is no exception. It treats a specific form of AMD, 'end-stage', a condition which affects about 500,000 Americans today over the age of 75. It is likely to increase significantly as the population ages. The IMT may not be able to treat all forms of blindness, but it is available now. Right now, in fact. If you have end-stage AMD you can talk to your opthalmologist about the implant today. That's very exciting news.
We've been following the IMT from VisionCare Inc. for a long time here at the Hub: we first covered the device more than a year ago. Last fall I had the opportunity to discuss CentraSight with Dr. Kathryn Colby, Professor at Harvard Medical School, surgeon at Massachusetts Eye and Ear Infirmary, and one of the lead investigators in the IMT's clinical trials. At that time she had been telling her prospective patients who wanted CentraSight that FDA approval could be arriving soon - a message she had been giving them for more than five years over the course of the FDA's review process. Now, those patients can sign up and seek the chance to improve their vision in a possibly life-altering way.
Will it be worth the wait? For most. But not for all. In their original 200+ patient study (published in the Archives of Opthalmology in 2007), Colby and her colleagues found that about 67% or more of those implanted with the CentraSight device saw notable improvement in vision. That's after one thirty five minute surgery.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


Of course it's also after many long months of rehabilitation and occupational therapy. The IMT doesn't treat AMD directly, it works around it. Incoming images are expanded onto the part of the macula that has yet to decay. Patients have to learn how to take that donut-like image and use it to see. If a patient has two eyes with AMD (which is often the case), one eye is kept un-implanted so that it can serve to pick up peripheral vision. It takes quite a bit of effort to adapt to this new kind of vision. Colby was quick to point out in our interview that not everyone with end-stage AMD will be a good candidate because of the stress of rehabilitation. There are also concerns with side effects such as corneal edema (swelling), and a small fraction, 1 out of 25 or so, will need a corneal transplant due to damage caused by the IMT. Yet for many over the age of 75, CentraSight represents a real opportunity to restore vision before it is lost completely. And as I've said, that opportunity is here now.
In the near term, VisionCare and their associated medical researchers like Dr. Colby will begin a new, larger clinical study as part of their post-FDA approval requirements. With 770 patients, the next CentraSight trial will further characterize parameters for success, and doctors will also be studying the long term risks on the original implant patients.
AMD affects about 30% of all Americans over 75 and VisionCare has estimated that more than a million patients will face end-stage AMD by 2050. When you include patients outside the US, those numbers swell. Of course, one hopes that by 2050 we'll have many more advanced options to treat all manner of blindness. Artificial retinas, I expect, should match or far exceed human vision by that time, and stem cells should be able to treat more and more varieties of blindness in the years ahead. It's great that CentraSight is now available to patients, but it will eventually be replaced by something better. One hopes that FDA approval has arrived early enough to allow patients to take advantage of the technique before their moment passes.
[image credits: Colby et al, Archives of Opthalmology, 2007, VisionCare]
[source: VisionCare Press Release, Illinois Eye and Ear Infirmary, Colby et al Archives of Opthalmology 2007]
Related Articles
Single Injection Transforms the Immune System Into a Cancer-Killing Machine

This Light-Powered AI Chip Is 100x Faster Than a Top Nvidia GPU

This Week’s Awesome Tech Stories From Around the Web (Through December 20)
What we’re reading