The Heart Rarely Gets Cancer. Scientists Think They Know Why.
The heart's constant motion makes it largely immune to cancer. The discovery could help protect other organs.
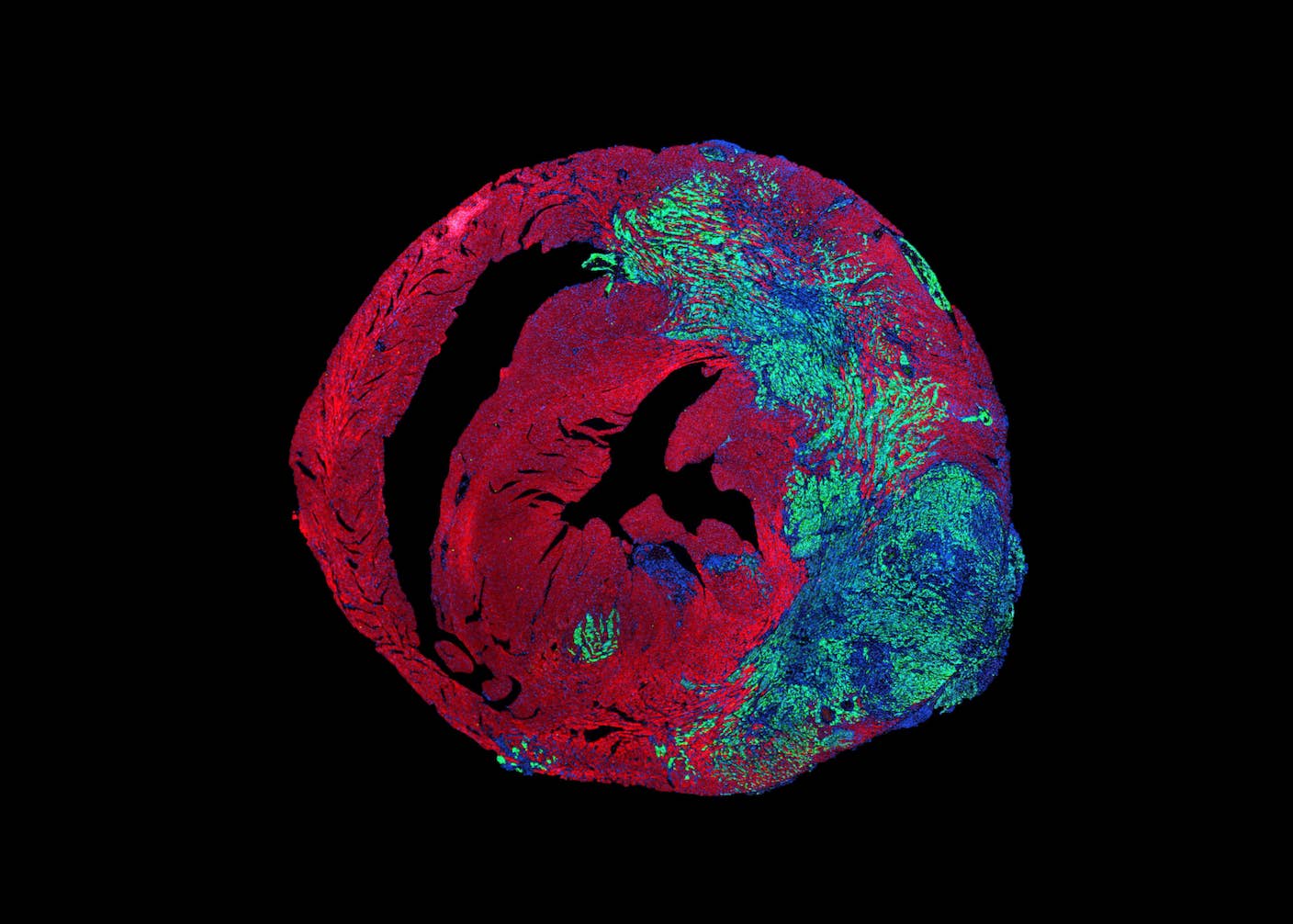
Image Credit
Ciucci et al., Science 2026
Share
The heart is a biological wonder. It beats roughly 2.5 billion times in an average lifetime. Unlike skin cells, which regularly die off and regrow, a healthy adult heart hardly regenerates at all—even through all the wear and tear.
The heart has another superpower: Resistance to tumors. Nearly every tissue in the body turns cancerous, but the heart almost never does. Cancers in heart tissue show up in less than 0.3 percent of autopsies, or about 1.5 cases per million people each year.
How the heart keeps cancer at bay has baffled researchers. Pinning down its hidden defenses could inspire treatments for more vulnerable tissues, including top killers such as breast, lung, or colorectal.
Persistent mechanical strain may be the key. A new study from the University of Trieste suggests that with every beat, the heart pushing against pressure dampens gene activity tied to tumor growth. In a rather Frankenstein experiment, researchers transplanted living hearts into the necks of mice, where they survived but didn’t experience mechanical stress.
When the team injected cancer cells, the mice’s own beating hearts slowed the invasion, while the transplanted hearts were nearly overtaken within weeks. Beating heart tissue grown in the lab also fought off tumors compared to tissue that didn’t beat.
Heart cells don’t uniquely feel stress. Lung, skin, and muscle cells do too, just in different, often less rhythmic ways. It’s possible that recreating heartbeat-like forces—potentially through wearable gadgets—could extend this type of natural protection to more common cancers.
Growing Pains
Cell growth is a double-edged sword. On the one hand, it’s essential for healing and regenerating the body. The skin is constantly blasted with radiation and toxins. It suffers cuts and bruises. To repair damage, skin cells turn over every 40 to 56 days. Bombarded with chemicals from food, medications, or alcohol, the liver’s cells regenerate to keep it in working order even after substantial injury.
But cancer is the price we pay for growth. Tumors arise as cell division damages DNA. Over time, cancers grow and spread. This is why we don’t get cancer in our teeth, nails, or hair—the cells making them up are dead. Cells that rarely divide also largely escape cancer. Mature neurons barely renew and seldom form cancers. Red blood cells, which lack a nucleus and DNA, can’t become cancerous at all. Heart muscle cells are similar. Despite nonstop contraction and damage, only about one percent or fewer renew themselves each year.
This partially explains why primary heart cancers or so rare. But the organ also wards off invading secondary cancers metastasized from other tissues, which are usually far more deadly.
“Even cardiac metastases are frequently clinically silent [no detectable symptoms], with many cases identified only incidentally or at autopsy,” wrote Wyatt Paltzer and James Martin at the Baylor College of Medicine, who were not involved in the study.
It’s a paradox. The heart is flooded with oxygen and nutrients, an ideal environment for wandering cancer cells to settle and thrive. Yet they don’t. One reason may be the heart’s inability to regenerate. Previous studies have suggested that the mechanical forces of heartbeats limit cell division. The team wondered if the same forces also shield the heart from cancer.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


Under Pressure
To test their idea, the researchers had to make a living heart with no beat.
“That was the most tricky part, because keeping the heart still is very difficult,” study author Giulio Ciucci told Science.
They adapted a technique used in end-stage heart failure patients to remove mechanical strain. In people, an implanted device takes over the pumping of blood. Here, the team transplanted a donor heart into a mouse’s neck and hooked it up to blood vessels. The animal’s own heart kept circulation going as usual. The transplanted heart stayed alive but didn’t do any work.
They then injected lung cancer cells, which often spread to the heart, into both organs. Within two weeks, nearly all healthy cells in the transplanted heart had been overtaken. In the beating heart, tumors rarely filled over 20 percent of a single chamber. Under constant pressure, the cancer cells struggled to divide.
One mouse with two hearts is hardly conventional. And transplantation risks immune attack and infection that could influence how cancers develop. “You have a lot of confounding factors,” said Ciucci.
So, the team moved to an “artificial heart” seeded with cancer cells, where mechanical forces could be dialed up or down in isolation. Like in the heart transplant results, the cancer spread throughout the tissue after removing strain. But it was mostly confined to the surface of beating tissues and in smaller amounts.
Looking for a reason, they compared gene activity in patient tissues with cancers that had spread to the heart, liver, and lungs and found a unique gene expression signature in the heart. In engineered tissues, mechanical stress changed how DNA was packaged, limiting access to genes related to growth and cancer. A protein on the surface of the nucleus, the cell’s DNA hub, translates physical forces from outside the cell into which genes are turned on or off. Knock this protein out, and invading cancer cells became “blind” to the heartbeat and grow freely.
Scientists have long known mechanical stress shapes cancer. As cancers grow, the cells stiffen surrounding tissue, which boosts survival, growth, immune evasion, and drug resistance. The new findings suggest that the movements of their host tissues also play a role, and the newly pinpointed protein could be a drug target.
The team is now exploring if mimicking heartbeat-like forces in other organs could prevent cancer growth. Lung, skin, and other tissues already stretch and relax, but remain susceptible.
“We really think that the key here is the continuous compression that you have in the heart,” said Ciucci. Working with engineers, they’re developing a wearable for melanoma—a type of skin cancer—that compresses the cells similar to a heartbeat. Early results look promising.
Dr. Shelly Xuelai Fan is a neuroscientist-turned-science-writer. She's fascinated with research about the brain, AI, longevity, biotech, and especially their intersection. As a digital nomad, she enjoys exploring new cultures, local foods, and the great outdoors.
Related Articles

AI Lab Partners Are Rewiring the Hunt for New Drugs

The Fully Anesthetized Brain Can Still Track a Podcast

Physicists Have Measured ‘Negative Time’ in the Lab
What we’re reading
