Computer Chip Implant to Program Brain Activity, Treat Parkinson’s

Share
An international team of researchers led by Dr. Matti Mintz at the University of Tel Aviv is working on a biomimetic computer chip for brain stimulation that is programmable, responsive to neural activity, and capable of bridging broken connections in the brain. Called the Rehabilitation Nano Chip, or ReNaChip, the device could be used to replace diseased or damaged brain tissue, restore brain functions lost to aging, and even treat epilepsy. The chip is currently in animal testing, but should reach human applications within a few years.
The ReNaChip will significantly improve an existing technology called deep brain stimulation (DBS), a surgical implant that acts as a brain pacemaker for a variety of neurological disorders. DBS delivers electrical stimulation to select areas of the brain via electrodes; for individuals with Parkinson’s, chronic pain, or dystonia, these induced stimulations can significantly alleviate symptoms (e.g. uncontrolled movement). But currently, the stimulation that DBS delivers is constant and unresponsive to brain activity. Because of this, the therapeutic effects are reduced over time. This is where the ReNaChip comes in, making the system responsive to brain activity and fully programmable.
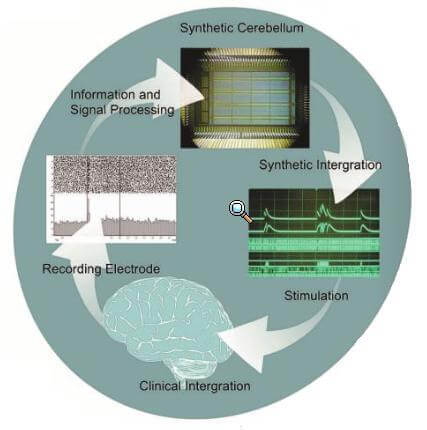
The key to the ReNaChip is that it is bidirectional – it deals in both electrical input and output. First, the system measures electrical signals that are normally present in particular neural tissue via electrodes implanted in the brain. These signals are transmitted to the silicon chip, which analyzes the signal with a variety of programmable algorithms. The chip then delivers electrical stimulation to an appropriate brain area along output electrodes. In contrast to current DBS technology, the ReNaChip would only deliver stimulation where and when it is needed (e.g. it could turn off when a patient is asleep).
Experimental studies are currently underway using rats as a model organism. For now, the researchers are applying the chip to a simple motor microcircuit in the brain: blinking, which is controlled in the cerebellum. The blinking microcircuit degrades with age, so current research aims to rehabilitate the response in aged rats. Input electrodes detect electrical impulses from cerebellar tissue, the silicon chip isolates the relevant signal from background noise, and an electrical stimulation is delivered to implanted electrodes that trigger blinking. If this proof-of-concept study is successful, they will move on to rehabilitating more complex neural wiring.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


While researchers are primarily focusing on motor responses, the applications of the ReNaChip are pretty wide. Any interrupted brain wiring (e.g. as a result of stroke) could conceivably be reconnected using electrodes and the flexibility of the chip’s programming. The chip could also be used to treat epilepsy, if electrodes could detect on oncoming seizure and diffuse it with appropriate stimulation. But researchers have their sights on an even more ambitious goal: rehabilitating the brain’s learning capacities, which would require increasing neural plasticitity. If the ReNaChip could be used to create and strengthen new connective networks, it could partially improve an older brain’s ability to learn new tricks.
There are a few problems with the ReNaChip as it exists now. The size of electrodes limits the precision with which signals can be recorded and delivered. An effort to further miniaturize the electrodes is underway, which would improve the larger DBS overstimulation problem as well. The actual chip size is also an issue of concern; researchers hope that someday the chip could be etched onto the electrodes themselves. Still, they don’t have any plans to put the chip itself inside the brain just yet – it would be inserted under the skin, much like a pacemaker. Dr. Mintz says the device will need about 6 more months of animal testing, and could reach humans within a few years.
The brain-computer interface is pretty hot territory right now, with research ranging disciplines from medicine to robots. We recently covered a monkey controlling a robotic arm via brain implants at the University of Pittsburgh, which gives a good idea of where the field is heading. But not all applications are so far out; next time you think that rewiring the body is mad science, consider how common and effective a simple cochlear implant is... and just to pull the heartstrings, here's a video of a baby hearing its mother's voice for the first time. As neural circuitry is more fully integrated into computer interfaces, we can expect more exciting research in medicine, neuroscience, and prosthetics.
The ReNaChip project is a collaborative effort between multiple international companies and institutions: Newcastle University, WizSoft Data and Text Mining, Universitat Pompeu Fabra, Lung University, Tel Aviv University, Guger Technologies, and Istituto Superiore di Santina.
[image credit: St. Jude Medical; North East Vision Magazine]
Drew Halley is a graduate student researcher in Anthropology and is part of the Social Science Matrix at UC Berkeley. He is a PhD candidate in biological anthropology at UC Berkeley studying the evolution of primate brain development. His undergraduate research looked at the genetics of neurotransmission, human sexuality, and flotation tank sensory deprivation at Penn State University. He also enjoys brewing beer, photography, public science education, and dungeness crab. Drew was recommended for the Science Envoy program by UC Berkeley anthropologist/neuroscientist Terrence Deacon.
Related Articles

AI Can Now Design and Run Thousands of Experiments Without Human Hands. We Aren’t Ready for the Risk to Biosecurity.

How Fast Are You Aging? New Genetic Clock May Have the Answer

An AI Solution to an 80‑Year‑Old Problem Has Shocked Mathematicians
What we’re reading