It is a villain that goes by many names: Type I, Juvenile onset, Diabetes Mellitus. More than 15,000 young people in the United States develop Type I diabetes each year, making them about twice as likely to die as their peers at every stage in their life. But there is a new superhero in the fight against Type I diabetes: Autologous Nonmyeloablative Hematopoietic Stem Cell Transplantation or AHSCT. While the name probably won’t fit on a cape, it has the potential for reversing some or all of the symptoms of Type I diabetes. Recently published work from Northwestern University in Chicago and the University of Sao Paulo in Brazil reveals new developments in this technique for using blood stem cells to treat patients, allowing them to go up to 4 years without needing insulin injections.
In Type I Diabetes, the body’s own immune system attacks Beta Cells in the pancreas. These cells help create insulin, a hormone which assists in the breakdown of sugar in the blood. Without this hormone, the body’s blood sugar levels are out of control, leading to a host of medical problems for the patient and often death if untreated. Treatments for Type I Diabetes consists of regular monitoring of blood sugar levels and the injection of artificial insulin, yet even with such treatment blood sugar levels are not optimal and health problems persist.
Dr. Richard Burt at Northwestern and Dr. J.C. Voltarelli at USP first reported their collaboration on using stem cells to alleviate diabetes in 2007. At that time they described the AHSCT process:
* Patients newly identified with having Type I diabetes were given drugs to stimulate the production of their blood stem cells.
* Those blood stem cells were harvested and frozen.
* The patients were given treatments that suppressed or destroyed much of their immune system cells.
* The patients’ own blood stem cells were reintroduced to their bodies.
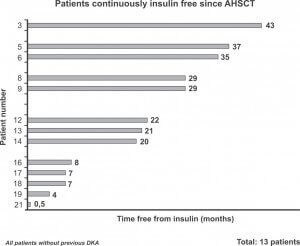
Now, in the April 2009 Journal of the American Medical Association (JAMA), Dr. Burt, Dr. Voltarelli, and their colleagues unveiled that patients in their study went up to four years without the need for outside insulin. The average time spent without the need for insulin was around 2.5 years. More than 90% of the patients showed some improvement. Of the patients that relapsed (started requiring insulin shots again) around 25% eventually recovered and regained insulin independence.
“At the present time (it) remains the only treatment capable of reversing type 1 diabetes mellitus in humans,” —the Voltarelli research group.
Every Superman has his Kryptonite
This isn’t Dr. Burt’s first time using stem cells as a cure, nor his first time in Singularity Hub. Our story on Crohn’s disease featured his work as well. In that case, as in this one, stem cell treatments were beneficial but not final. As remarkable as the AHSCT’s treatments are, they can only last so long before needing to be repeated or replaced with another therapy.
AHSCT’s pioneers readily admit that is far from a cure for diabetes. Patients have to be identified very early or Beta cell depletion may already have progressed too far. For millions already suffering from Type I diabetes, then, AHSCT holds no likely benefits. This problem may be solved if a new and readily available source of blood stem cells could be found for each patient. Whenever it is used, however, AHSCT will be an expensive and difficult treatment, requiring long hours of work at a specialized facility.
“Randomized controlled trials and further biological studies are necessary to confirm the role of this treatment in changing the natural history of (the disease),” — the Voltarelli research group
AHSCT also carries some dangers. There are risks for complications in both the short and long terms. The treatment weakens the immune system for a time. In this study, two patients developed pneumonia, three had endocrine dysfunctions, and nine developed oligospermia (low sperm count). There were no mortalities, but these risks cannot be ignored.
Dr. Jay S. Skyler, from the University of Miami, has risen as an optimistic critic of the work. In a related 2007 editorial he points out several key flaws in the trial:
*There was no control group.
*Remission is common in the first months and years following diagnosis of Type I diabetes.
*It is still too soon to tell if the technique really works in the long term.
*It is unclear how the technique actually works (if it protects beta cells or allows them to regenerate).
While Skyler and many others in the world of diabetes research don’t want to call AHSCT a cure, they are hopeful that this work will eventually lead to a successful reversal for the disease. Voltarelli and Burt admit that the eventual solution for diabetes may come from an entirely different direction such as chemical treatments, umbilical cord cells, adult stem cells, embryonic stem cells, or immunoregulatory treatments at the cellular level.
“As these further studies confirm and build on the results of Voltarelli and colleagues — the time may indeed be coming for starting to reverse and prevent type 1 diabetes mellitus,” —Dr. J.S. Skyler
Whether or not AHSCT becomes a cure, it may stand as a model for other treatments of illnesses related to the immune system such as AIDS or Lupus. Certainly the process of reintroducing stem cells has as many applications as there are functions of stem cells. At the moment, AHSCT is remarkable simply because it shows a solution to these types of diseases is at least possible. It’s hard to accept that our heroes, or our cures, may have their flaws. But that doesn’t prevent them from providing hope.


