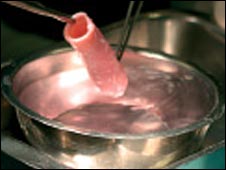
Two very different organ transplant stories highlight the amazing skills of surgeons, and the importance of stem cells for the future of surgery. Both transplants involved the trachea, women, and operations in Europe. In the first, Claudia Castillo had a donor’s trachea covered in her own stem cells so that it could be transplanted into her chest as the left bronchus. The second transplant gave Linda de Croock a new trachea by first letting the donor organ acclimate to her body in her arm for ten months. The stem cell treatment was performed by doctors at the University of Bristol (UK) and the Hospital Clinic of Barcelona (Spain) in 2008. The trachea-in-arm treatment was performed at the University Hospital of Leuven (Belgium) and was recently discussed in the New England Journal of Medicine. Comparing these two operations demonstrates the phenomenal capabilities of modern medicine, and shows that stem cells enhance those capabilities in remarkable ways.
Excuse me, is your arm coughing?
Linda de Croock, now 54, lived for 25 years with metal stints in her throat to keep her windpipe open. Her own trachea had been crushed in a tragic car accident. According to her conversations with journalists the stints were horribly painful, affected her ability to communicate, and lowered her quality of life. She sought out Dr. Pierre Delaere, whom she discovered via the internet, and who had performed impressive transplants for cancer patients. The concept that Delaere and his colleagues developed was simple: to reduce the chance of rejecting a donor trachea, they would first wrap the organ in de Croock’s own cheek cells and acclimate it to her body by resting it in her arm.
Similar work had been done for many other organs, including using the muscle tissue of the back to prepare parts of a jaw. Yet this trachea-in-arm maneuver would be the largest organ to ever undergo such treatment. de Croock kept the trachea in her arm for ten months, with layers of plaster around the limb to keep it safe. She also was kept on a steady regimen of immunosuppressants to prevent rejection. After four months, mucous was growing healthily on the organ. Nearer the ten month mark, suppression therapy stopped and the organ remained healthy. Eventually, the chimeric trachea was transplanted into the throat. Once there, no further immunosuppression therapy was needed.
Linda’s new trachea is doing well. She can speak much more easily now, and her breathing is normal. Because she doesn’t require any further suppression therapy, she is not as vulnerable to illness, nor runs the increased risk of cancer associated with those medicines. Doctors were so pleased with de Croock’s results that they performed the same surgery on an eighteen year old male and have two more patients in queue.
I would just like everyone to digest that for a moment. Doctors wrapped a dead man’s trachea in cheek tissue, stored it in an arm, and got it to successfully transplant into a throat. Not once, but twice. And they’re going to keep doing it, presumably with great success. Science and modern medicine blow my mind.
I’ll be back in a few weeks… with a new organ
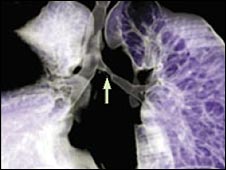
Tuberculosis damaged Claudia Castillo’s lungs and airways. The Colombian mother had such trouble breathing that she was often unable to play with her children or climb stairs. Her left bronchus was narrowing dangerously and some sort of surgery was needed. That’s where Professor Paolo Macchiarini in Barcelona and Professor Martin Birchall in Bristol stepped in. Previous work with pigs had shown that an organ could be stripped down using enzymes and other chemicals. This would leave the organ as nothing but a scaffold. Stem cells could be applied to this scaffold and encouraged to regrow the organ. Singularity hub has discussed similar work done with rat and pig hearts.
Macchiarini and Birchall were willing to try the procedure with Castillo and a donor trachea. The trachea was stripped down into a scaffold. Castillo’s stem cells were harvested from her leg bone marrow, and non-stem cells were taken from her throat. Birchall used these cells in a bioreactor in Bristol to regrow the scaffold in Bristol. In just four days, the organ was complete and because Castillo’s cells were used in the process, it would be recognized by her body and not rejected. Macchiarini then performed the operation to replace her damaged left bronchus with the new stem cell trachea.
Four days after that surgery, the team found that the new organ was virtually indistinguishable from the rest of Castillo’s body. After a month, tests proved that the transplanted trachea had developed it’s own blood supply – it was a viable part of her system. Four months after her treatment, Castillo’s ability to breathe showed great signs of improvement: she could climb two flights of stairs and take care of her children.
Again, a moment. Doctors used a stem cell technique to grow a (partially) new bronchial tube that was recognized as part of the patient’s own body. It only took a few days for that tissue to be assimilated and a few months for that assimilation to be (essentially) complete. My mind is blown. Again.
I’ll have what she’s having
If you need an experimental organ transplant, I’m sure that both of these stories are promising and uplifting. Yet I don’t think it’s disrespectful to the work done in Belgium to say that I would greatly, greatly prefer the stem cell treatment option. Who wouldn’t. It took four days to grow a new organ on a scaffold. Four more days before doctors were amazed at the organ’s acceptance after surgery. Jump ahead four months and Castillo is playing with her kids and climbing stairs (probably not at the same time, as that’s dangerous, but still). If these two procedures had been started at the same time Castillo would be mostly finished while de Croock still had months left incubating a trachea in her plastered arm. From a patient point of view, there’s almost no comparison to these two procedures.
Yet the Belgian team is moving forward quickly, racking up successes while, according to BBC News, the British/Spanish team hopes for clinical trials in similar cases to start by around 2013.
The discrepancy between the two stories isn’t caused by stem cell controversy. There’s no political hold up, and there’s not a lack of funding. No private interest groups are fighting over intellectual property rights. None of the commonly debated impediments are keeping us from enjoying this new and promising surgery.
It’s just science. The trachea-in-arm technique may seem like it came from the mind of Dr. Frankenstein, but it is a lab tested, hospital approved, and proven means of prepping organs for transplants. There are years of clinical trials and human lives saved behind this technique. Admittedly the size of the trachea is formidable, and larger than previous organs, but it’s the same basic idea. Stem cells scaffolding for whole organs is still relatively new. It’s mostly in the lab phase. There has to be years of steady tests and experimental procedures like this one before doctors are certain that it’s a good treatment for patients.
But that’s also the really hopeful part about the Castillo transplant. The only thing standing in its way is the regular (and necessary) progression of science. All signs point to the scaffolding technology as developing into a very successful alternative/compliment to organ transplants. Scaffolding for bladders and simple tissues are proceeding very well in the United States, moving into Phase III of trials. As they move forward, so too will more complex organs like tracheas, and hearts. Give medicine a decade, and such techniques may be the norm. In the meantime, the high quality and inventiveness of surgeons will produce techniques like the de Croock transplant. Some of those techniques will live on as stem cells aren’t always an option for treatment. It’s a win-win. Frankenstein for now, stem cells in the near future. Medicine is cool.
[photo credits: BBC News]


