Programmable Bio-Nano-Chips: First Viable Medical Lab on a Chip?

Share
We often find out too late what's happening inside our bodies when it's stricken by disease or cancer, but those days could be coming to an end. The McDevitt Lab of Rice University has developed a Programmable Bio-Nano-Chip (PBNC), an alternative to the costly, time-consuming, and sometimes painful procedures for diagnosis. By using nanoscale fluorescent labeling to look for biomarkers linked to disease, these credit-card size chips could enable physicians to recognize heart disease or cancer with just a small saliva sample, no catheterizations or biopsy required. PBNCs also represent an exciting milestone for the “lab-on-a-chip.” In the past, this technology could analyze very small samples, but it has been economically impractical and still required a full-scale laboratory. PBNCs don't have these issues. Chips for various conditions are already in clinical trials, so the McDevitt team’s PBNC is quickly forging a path to a facility near you. The arrival of this technology would enormously benefit the healthcare system by improving clinical outcomes and reducing costs. PBNCs also hint at other possibilities, like detecting the earliest signs of disease. If these devices ever become implants, then the Body 2.0 upgrade we’ve been waiting for could be within reach. Your body's health, right down to its biochemistry, could be as easy to read as your wristwatch.
PBNCs fit the functionality of a diagnostic laboratory on a small silicon chip. Rice University developers applied fluorescent antibody labeling (FAL), a technique in which fluorophore-tagged antibodies attach to specific unbound or cell membrane proteins in order to determine concentration. The team miniaturized this method so that it could detect these protein biomarkers down to a few billionths of a gram. This allowed the device to discern when certain indicators of pathology (i.e. abnormal concentrations or uncontrolled cell proliferation) appear in the body, using only a saliva sample. When the McDevitt lab combined miniaturized detection with microfluidics, a stand-alone reader, and a reprogrammable sample processing core, the PBNC was born.
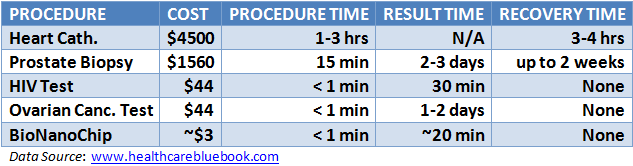
The video below highlights the functionality of the device as well as its potential fiscal benefits. At 1:26, Dr. McDevitt describes the grim situation of conventional heart disease diagnosis, pointing out that too often “the first symptom they get is they die.” Then, a lab member divulges the technical details of the PBNC-based diagnostics (1:55). He shows a case of a test performed on a man who was complaining of chest pains, with the nine central beads emitting more fluorescence compared to a non-heart attack patient. This indicated an elevated concentration of cardiac troponin I, a revealing biomarker for cardiovascular damage. The video wraps up with Dr. Vivian Ho (4:32) describing how PBNCs conflict with the dogma of healthcare economics: better technology inevitably leads to rising costs. However, according to Dr. McDevitt, mass chip production could drive the cost of PBNCs down to a few dollars per test. Compare that to other diagnostics in the table below the video. If PBNCs lead to both better health and less financial strain, we could have our cake and eat it too.
So when will these tests reach hospitals and clinics? According to Dr. McDevitt:
More than 20,000 scientific-discovery papers for cancer and 6,000 for cardiac disease have been published, yet only about one biomarker per year for all diagnostics areas received FDA approval from 1995 to 2005.
While regulatory protocol has impeded biomarker-based testing in the past, Dr. McDevitt’s promising technology has prompted the FDA to set PBNCs on the fast-track for approval. Chips for heart disease and three different kinds of cancer are already in Phase III clinical trials. For those unfamiliar with the FDA approval process, Phase III is one of the last steps before drugs or medical devices are allowed to be marketed. Barring any significant issues during the multi-site trials, it appears PBNCs could be just around the corner.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


If these devices ever become widespread, I wonder what other disorders these chips will be able to detect in the future. The biomarker signatures for many diseases are still being worked out, but once they are found, it’s not unreasonable to assume that PBNCs could be reprogrammed for them as well. For example, there has been headway using biomarkers to detect neuropsychiatric dysfunction. Currently, the blood test for schizophrenia costs around $2500, so a PBNC version of this test could make it much more accessible. One issue to consider is that a PBNC for schizophrenia would need to assess 51 biomarkers, and the PBNCs in clinical trials right now only quantify three or four. While there will be logistical concerns if these chips expand to testing other diseases, I’m confident that these challenges will be surmountable.
Looking even further ahead, could PBNCs ever be integrated with our biology? We’ve seen cyborg retinas, a silicon pancreas, and a vitals clip-on, but what can PBNCs offer to Body 2.0? Not much . . . for now. The chips are exclusively body-external for both sample collection and analysis. However, reflecting the general trend in microelectronics, I would expect that PBNCs will continue to miniaturize. After confronting issues with biocompatibility to ensure the body does not reject the device, then PBNCs could be surgically implanted. The chips would constantly monitor the levels of biomarkers for any disease imaginable. It could also be combined with a peripheral transmitter that relays biometric data to a receiver outside the body for analysis. Such an apparatus would enable us to detect disorders as they emerge in real-time, the pinnacle of preventative medicine. While this might sound like a medical pipe dream, there is considerable interest in implantable biochips. In fact, this technology has received big-time government funding in the past.
I am very excited about this technology and hope the FDA places similar tests on the fast-track of approval. Like countless others out there, cancer has been a scourge of my family. Getting a biopsy, waiting for the results, and paying for the costly lab tests can take a toll. While waiting for any life-determining test result isn’t easy, I would much prefer a 20 minute delay and avoid a painful biopsy. As PBNCs make their way through clinical trials, I will be watching the headlines, patiently waiting for the day that the grueling diagnosis process gets a little bit easier.
To learn more about PBNCs and how it could impact global health, watch this in-depth presentation by Dr. McDevitt given at Berkeley.
<Image Credits: Screenshot of Rice University News and Media Relations PBNC Video>
<Video Credit: Rice University News and Media Relations>
<Sources: Rice University News and Media Relations>
Related Articles
MIT Mined Bacteria for the Next CRISPR—and Found Hundreds of Potential New Tools

US Issues Grand Challenge: The First Fault-Tolerant Quantum Computer by 2028

This Week’s Awesome Tech Stories From Around the Web (Through April 4)
What we’re reading