Flu Vaccine Produced With Insect Ovaries Approved By FDA – The End To Shortages?

Share
Amid a season in which the flu is spreading at an exceptional rate across the United States, the Food and Drug Administration has approved a vaccine that can get to the people who need it quicker than conventional vaccines.
FDA approval came January 16 for the new vaccine, called Flublok, which was developed by Protein Sciences. It differs from conventional vaccines in that its production process does not involve an influenza virus nor growth in eggs. By a unique process involving insect cells, Flublok production begins after three weeks. Conventional vaccines normally take four to six months to begin production.
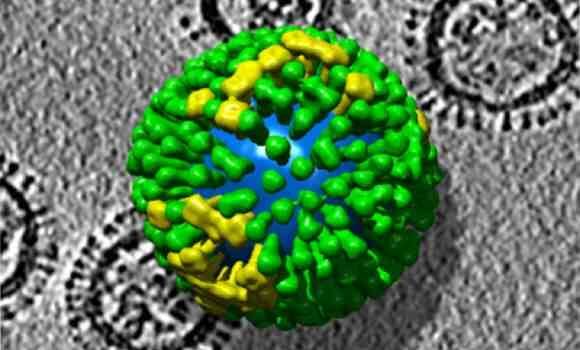
Hemagglutinin (HA) is a protein found on the influenza virus that enables it to enter and infect cells. It is also the protein that makes the flu a moving target. Vaccines that protect against infection contain antibodies that target HA. But with each new flu season new strains appear in which their HA sequence has been modified. This is why new vaccines have to be made each year with antibodies that recognize the new HA sequence.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


Historically the new vaccines are created by first culturing viruses in fertilized chicken eggs and, more recently, in cell cultures. The cultured viruses are then extracted from the eggs, broken up and their HA proteins purified. Alternatively, Flublok is developed from DNA containing only the sequence for the relevant HA proteins. This DNA is incorporated into DNA from a virus that only infects the cells of insects. So, for Flublok, the researchers proliferate the viral DNA in ovaries taken from the fall armyworm moth.
Flublok recognizes three of this season’s most prevalent flu strains. In a phase III study Flublok was shown to protect people against 44.6 percent of all flu strains, not just the three strains against which the vaccine was matched.
FDA has approved the vaccine for people between 18 and 49 years of age. According to Manon Cox, chief executive of Protein Sciences, 150,000 doses of Flublok could be made available before the end of this flu season. While that’s not a lot of vaccine, the fact that chicken eggs are not involved in the production process means the vaccine could help those who are prevented from getting flu shots due to allergies to eggs.
Flublok is the result of an overt FDA goal to reducing flu vaccine lead times and increase capacity. Recently a new method was developed in Germany that used messenger RNA for quicker production. At $30 a dose Flublok is slightly more expensive than conventional vaccines. But its quicker time to distribution means the extra expense might be worth it, not just for individuals but for a society that is much better equipped at containing the flu before it becomes widespread.
Peter Murray was born in Boston in 1973. He earned a PhD in neuroscience at the University of Maryland, Baltimore studying gene expression in the neocortex. Following his dissertation work he spent three years as a post-doctoral fellow at the same university studying brain mechanisms of pain and motor control. He completed a collection of short stories in 2010 and has been writing for Singularity Hub since March 2011.
Related Articles

Industries Most Exposed to AI Are Not Only Seeing Productivity Gains but Jobs and Wage Growth Too
One Shot Just Crushed Three Deadly Autoimmune Diseases

Scientists Grow Electronics Inside the Brains of Living Mice
What we’re reading