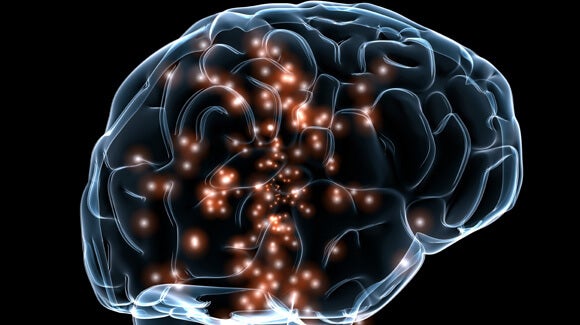
Massive Military-Funded Project Aims to Re-align Ailing Brains

Share
Deep brain stimulation as a treatment for epilepsy and movement disorders, most notably Parkinson’s disease, has rapidly gone from experimental to standard practice. With devices to provide delicate electro-stimulation to the brain now available and with maps of which neurons do what steadily gaining detail, attention is now shifting to using the approach to treat mental illness.
Existing medications and psychotherapy work for just four out of five people who suffer from depression or anxiety. Anxiety disorders alone account for a full third of the U.S.’s national health care costs.
DARPA has just funded a massive research effort involving several universities, hospitals and private companies to explore the use of implanted electro-stimulators to treat depression, anxiety, traumatic brain injury and post-traumatic stress disorder. These last two are common problems in veterans returning from combat in Iraq and Afghanistan.
Researchers will monitor and record neuronal activity to identify normal patterns and those that suggest mental illness. They will then fine-tune implantable stimulator devices to stimulate the healthy patterns in people with mental illness, spurring their brains to develop stronger connections along those pathways. By the end of the five-year project, the devices should be ready for human trials.
The hope is that by teaching the brain to use normal pathways, brain stimulation could effectively cure patients of mental illness. Ideally, the implants would eventually be removed.
“This can be a big leap. We’re developing an entirely different approach to the treatment of mental illness by using brain implants to target a small population of neurons involved in neuropsychiatric disorders instead of using drugs that change broad swaths of activity,” said Jonathan Wallis, a psychologist at the University of California Berkeley who will also participate in the project.
With $26 million in funding, the project claims about an eighth of the research funding so far allotted by President Obama’s BRAIN initiative.
So who will volunteer to have a low-powered electronic device implanted in their brain to treat a disease with other treatment options and which isn’t generally fatal? The participants are patients already receiving deep brain stimulation to treat Parkinson’s disease or epilepsy.
Although they take a back seat in public conversations about the diseases, psychiatric symptoms are commonly features of both. In Parkinson’s disease, psychiatric symptoms ebb and flow, which will help researchers differentiate normal and abnormal brain activity in recordings in the absence of a control group.
The recordings are, themselves, groundbreaking: Scientists have never before been able to see what happens in a patient’s brain during episodes of mental illness. That data, minus the patients’ identifying information, will be made open source.
Although the research is cutting-edge, to be sure, the researchers aren't just fumbling in the dark.
“We know many of the parts of the brain that are really important and play critical roles in these disorders. But what we don’t understand is how these parts of the brain talk to each other and how they interact in ways that contribute to these disorders,” Vikaas Sohal, a psychiatrist at UCSF, the project’s hub, said in a video.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


Identifying physical signatures diseases as amorphous as depression and anxiety will help doctors arrive at the right diagnosis more quickly. Researchers will also be looking to see if some neurons play a part of more than one mental illness, which could open up new treatment models.
But developing low-power, miniature, wireless electronic devices that can see at the level of neurons is just as ambitious a part of the project as learning to distinguish, say, sorrow from depression. That work will be led by the Center for Neural Engineering and Prostheses, a joint project of the San Francisco and Berkeley campuses of the University of California.
“In this new project at CNEP, we are using the same BMI concepts we have been using for the past 10 years. After learning how large-scale brain circuits work in such conditions as depression, anxiety, and addiction, we will design an implantable BMI that can detect abnormal activity and electrically stimulate some locations in these circuits to alleviate symptoms,” said Jose Carmena, an expert in brain-machine interfaces at the center.
Much as neural activity related to motion can drive a robotic prosthetic, it could also send commands to the stimulators to activate the healthy parts of a patient's brain. The treatment would be able to adapt as the patient improves or otherwise changes.
Researchers expect to use equipment from Medtronic, maker of the Parkinson’s brain stimulator, the Lawrence Livermore National Laboratory, Cortera Neurotechnologies and the Cambridge, Mass.-based nonprofit manufacturer Draper Labs.
Images: DARPA, Draper Labs, Medtronic
Cameron received degrees in Comparative Literature from Princeton and Cornell universities. He has worked at Mother Jones, SFGate and IDG News Service and been published in California Lawyer and SF Weekly. He lives, predictably, in SF.
Related Articles
New Algae Robots Swarm Like Locusts at the Flick of a Switch

In the Scramble to Power AI, Investors Bet $140 Million on Data Centers at Sea

Robots With Different Designs Can Now Share Skills
What we’re reading