The Radical Implant That Enables Paralyzed Rats (and Maybe One Day Humans) to Walk Again

Share
Scientists at the Ecole Polytechnique Fédérale de Lausanne (EPFL) in Switzerland recently unveiled a flexible spinal implant called the electronic dura (or e-Dura) that they say may greatly improve spinal injury rehabilitation. In a paper published earlier this month in the journal Science, the EPFL team said because of its flexibility, the next-generation implant lasts longer (up to two months) and causes much less damage than traditional implants.
These latest results continue a line of research into regenerative medicine by EPFL scientists led by Dr. Grégoire Courtine and Dr. Stéphanie P. Lacour. In 2012, this same group published stunning results showing rats with paralyzing spinal cord injuries were able to regain the ability to walk, run, and even climb.
When the spinal cord is severely injured, it results in loss of control over the part of the body below the point of injury.
The EPFL team learned that an injured spinal cord could be reactivated in rats with a specific combination of drugs and electrical stimulation. The drugs, called monoamine agonists, activate the same receptors as neurotransmitters in healthy subjects. Exposing the injured spinal cord to these drugs, along with mild electrical stimulation, activated nerve cells in the spinal column and produced movement in the paralyzed animal.
Early on, the brain still wasn’t communicating with the area below the injured spinal cord, and the movement was involuntary. Over time, however, as the animals (supported by a robotic harness) gained confidence in their ability to walk again, the scientists noted a fourfold growth of new nerves in the spinal cord—the nerve growth eventually restored communication between the brain and the injured area of the spine.
As you can imagine, the researchers were eager to take these findings to the clinic to see if they might have the same success with humans. In preparation for clinical trials, however, they came up against another hurdle—the need for a long-term spinal implant to deliver the chemical and electrical stimulation needed to initiate healing.
They hope e-Dura can satisfy their needs.
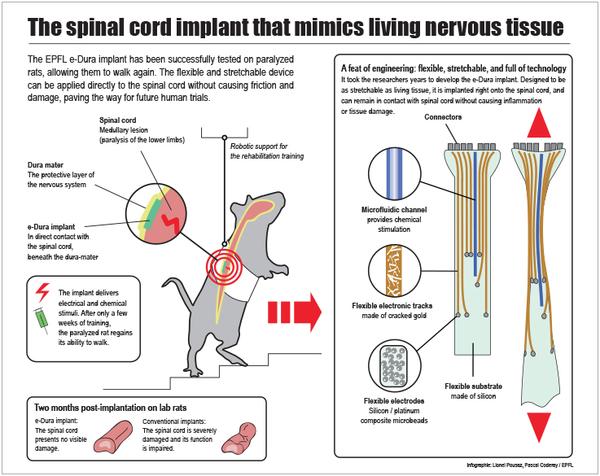
The device meets two important criteria for spinal implants: durability and biocompatibility. To reduce the number of surgical procedures that an injured patient needs to undergo, an implantable device needs to last a long time. And it needs to be biocompatible and flexible. Early generation implants caused side effects like inflammation and scar tissue, often negating any positive results the implant provided.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


In testing on laboratory animals, the researchers applied the e-Dura implant beneath the protective dura matter, directly on the spinal cord. The implant did not cause any adverse effects and lasted long enough for the paralyzed animals to complete their rehabilitation.
Functionally, the implant also performed very well.
Tiny microfluidic channels embedded on a flexible silicon substrate deliver precise amounts of drugs directly to the nerve cells. Cracked gold conducting wires and electrodes made from a silicon and platinum composite send electrical signals to the injured spinal cord. The implant’s electronics also allow the researchers to monitor the electrical messages sent back and forth to the brain as the new nerves are activated.
Although the research is very exciting, it is yet unclear how well it will work in humans.
It’s possible humans will need different drugs or a different pattern of electrical stimulation to coax the spinal cord into healing itself. There are many unknowns. Advanced clinical trials will hopefully elucidate these nuances so that an appropriate formula can be determined for human applications.
While exoskeleton technology also continues to approach consumer markets, it would be better to give people their natural ability to walk back. However you approach it, those suffering paralysis due to spinal injuries may have more options in the coming years.
To learn more about this incredible research, check out Dr. Courtine’s TED talk from June 2013.
Related Articles
One Shot Just Crushed Three Deadly Autoimmune Diseases

Scientists Grow Electronics Inside the Brains of Living Mice
How to Build Better Digital Twins of the Human Brain
What we’re reading