New Reports Show ‘Artificial Pancreas’ Is On Route To Treat Diabetes
Share
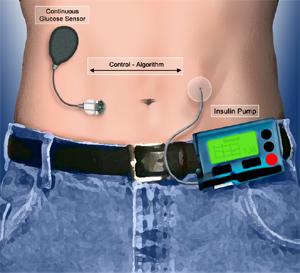
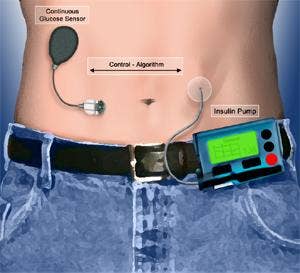
An 'artificial pancreas' developed with funding from the Juvenile Diabetes Research Foundation (JDRF) could allow diabetics to control their blood sugar automatically. Consisting of an insulin pump, continuous glucose monitor, and advanced control software, the artificial pancreas is part implant and part worn device. It will monitor blood sugar and vary insulin levels without a patient's need for constant calculation and adjustments. According to recent presentations at the American Diabetes Association Scientific Sessions in Florida, the artificial pancreas has shown great aptitude for treating diabetes overnight and after large meals - difficult times for many diabetics. The device may prove to be the best treatment for diabetes until biological based solutions are completed.
Diabetes is one of the most widespread and costly diseases in the developed world. Linked to the growing obesity epidemic in the US, about 8% of the country has the condition, and its the seventh leading cause of death in the nation. The bodies of diabetics have difficulty controlling insulin levels in the blood, often due to malfunctioning cells in the pancreas. There have been successful attempts at getting stem cells therapies to treat diabetes. While these therapies may prove to be actual 'cures' for the condition, they are unlikely to be perfected or widely available for many years. The JDRF, by pursuing an artificial pancreas is essentially looking to provide a quicker solution to managing diabetes.
The artificial pancreas essentially combines two already well researched technologies. The first is the insulin pump, an externally worn device with a tube connected directly into the abdomen which allows patients to add insulin (or glucagon) into their blood stream without using syringe injections. The second technology is the continuous glucose monitor (CGM) which does exactly what its name suggests: continually track blood sugar levels and inform its user when an injection is likely to be needed. The CGM can effectively replace the standard prick-your-finger blood tests most diabetics rely upon. In the artificial pancreas, the CGM is an internal implant which communicates with the outside world via wireless signals. It actually measures glucose levels in the interstitial fluid rather than the blood itself. Bridging the two technologies is a sophisticated software package that translates data from the CGM into commands for the insulin pump. It's this software, and the related hardware communication innovations, that really set the artificial pancreas above previous devices. With the right code, the insulin pump and CGM can work in tandem effectively regulating a person's blood sugar within desired parameters for almost any condition.
There aren't many videos of the artificial pancreas in action, and unfortunately most are old. The following clip from CBS gives a fairly good overview of the technology. Newer, though not necessarily more informative, videos can be found on the JDRF website.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


JDRF's artificial pancreas has enjoyed several successes this year. The first was recorded in the Lancet, as Roman Hovarka and colleagues at the University of Cambridge demonstrated it was equal or better than standard treatments when regulating blood sugar in normal conditions, after exercise, and after large meals. Recent presentations at the ADA suggest that the artificial pancreas is also much better in overnight regulation of blood sugar. Patients staid within desired levels 70% of the time for the artificial pancreas versus just 47% of the time using standard routines. Also presented at the ADA Scientific Sessions was a JDRF funded psychology study on who would likely benefit from and successfully use a CGM. The results from that study (such as teenagers are unlikely to stick with CGM use) will be helpful not just in determining which groups are prime targets for using the artificial pancreas, but in shaping public health decisions in how to get patients to adopt newer diabetes technologies that could save money and lives.
Development of the artificial pancreas has been divided among many different institutions including Yale University, Cambridge University, Harvard Univesity, University California Santa Barbara, and several others - all funded by the JDRF. Furthermore, the project is compatible with several different types of insulin pumps and CGMs already on the market. The diversity in research and commercial devices is a big strength of the project - increasing the chances that an artificial pancreas may actually be a marketable item in the next two to five years (the figure often quoted in press for the JDRF). We should keep in mind, however, that studies into its work are still limited in size. The Lancet research involved just 17 people and not all were tested in each area of interest - large meals, exercise, normal use. This is a promising project, and it may arrive in the next few years, but only if it can show success in several larger trials.
The JDRF, ADA, and other diabetes groups are open about the limitations of an artificial pancreas as seen today. It's essentially seen as a stop-gap solution to treating diabetics, providing a solution by automating an approach (monitor blood sugar, add insulin as needed) that has been around for decades. Ultimately we'll want an entirely new way to treat diabetes - most likely stem cell therapies which restore the body's natural ability to regulate insulin. Genetic research may finally pinpoint the causes for the condition and start treatments early. We should also remember that while diabetes has a genetic component, healthy living styles (eating less, exercising more) can have a positive effect and may help us fight diabetes (and obesity) as a population. As we wait for those techniques to be perfected and adopted the artificial pancreas has the potential to help diabetics live with less concern and more freedom. Let's hope it continues to see successes in the years ahead.
[image credit: JDRF]
[video credit: CBS]
[source: JDRF, Hovorka et al, Lancet, ADA statistics]
Related Articles

The Fully Anesthetized Brain Can Still Track a Podcast

This Week’s Awesome Tech Stories From Around the Web (Through May 16)
New Algae Robots Swarm Like Locusts at the Flick of a Switch
What we’re reading