Drug To Diagnose Alzheimer’s Disease Receives FDA Approval

Share

Amyvid, a drug that binds to a marker for Alzheimer's disease, gives physicians a new tool with which to differentiate between normal (left) and Alzheimer's (right) individuals.
Pharmaceutical giant Eli Lily just got FDA approval on a chemical that would enable clinicians to detect a biological marker for Alzheimer’s disease. They can detect the marker now, but currently quantifying it can only be performed during autopsy. Detecting it early, during a person’s lifetime has the potential to not only identify people at risk for Alzheimer’s before they show symptoms, but it could help researchers searching for a cure.
The chemical, called florbetapir or its brand name Amyvid, binds to the protein, beta-amyloid, thought to be a hallmark of Alzheimer’s disease. The drug, which is radioactive, is injected into patients which are then imaged with a Positron Emission Tomography (PET) scan that detects the radioactive signal. A positive scan means there are at least a moderate amount of amyloid plaques, the aggregates of amyloid protein thought to disrupt neuronal function and lead to cognitive decline and dementia associated with Alzheimer’s disease.
Right now Alzheimer’s disease is diagnosed by individual physicians who detect cognitive and behavioral changes in the patient. But by the time behavioral changes are evident, scientists think, the disease is already very advanced. They think that the disease actually begins years before symptoms start to show. If there was a way to detect the disease, such as a PET scan that detects an increase in amyloid protein, doctors could identify high risk patients years before they begin to show symptoms.
Unfortunately, that won’t help the patients very much as there is currently no cure for Alzheimer’s disease. But the real strength of the test rests in its ability to differentiate Alzheimer’s patients who have amyloid with those who do not.
Between 10 and 20 percent of patients diagnosed with Alzheimer’s disease do not show abnormal levels of amyloid at autopsy. The cause of the disease may be different in these patients than in the patients with elevated levels of amyloid, and thus different treatments may be required. Conducting separate studies on these two groups could be a better way to finding a cure.
Researchers also estimate that in some communities a third of patients with mild symptoms but nonetheless have Alzheimer’s disease go undiagnosed. If these patients also show increased levels of amyloid doctors may be quicker to the diagnosis.
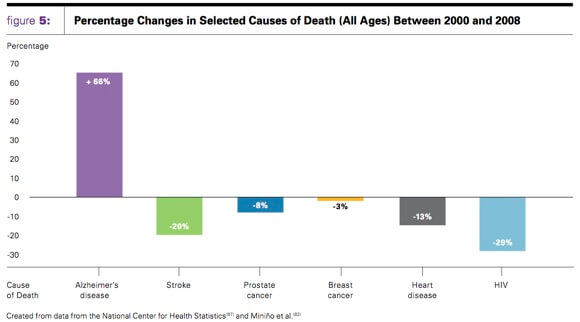
Deaths due to Alzheimer's disease continue to rise while falling for other major diseases (source: Alzheimer's Association).
Research seeking treatment for Alzheimer’s also stands to benefit from detecting amyloid earlier. Nowadays people have to already show signs of cognitive decline to qualify for clinical trials. The problem is, the damage is already done by the time outward symptoms begin to show. If it turns out that the amount of amyloid increases appreciably before they show cognitive decline, these people at risk to develop Alzheimer’s could be enrolled in clinical trials earlier. And clinical trials aside, just correlating the timing of amyloid increase to the onset of behavioral symptoms could help researchers understand how the two are related.
Amyvid adds to the recent growth of the Alzheimer’s diagnosis toolkit. Tests, shown to be extremely accurate in differentiating Alzheimer’s and normal individuals, are already commercially available to measure amyloid from spinal fluid. A bit less traumatic than a spinal tap, a blood test was developed last year that correctly identified over 80 percent of Alzheimer’s patients based on the levels of nine hormones and proteins including amyloid. And GE Healthcare is currently developing another PET approach that uses a different tracer to bind amyloid, [(18)F] Flutemetamol. Phase III trials for the drug are underway.
Eli Lily said Amyvid will be available this June in “limited quantities.” Side effects from the drug include headache, fatigue, muscle pain, and nausea.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


Alzheimer’s disease affects 54 million Americans and is the sixth-leading cause of death in the US. It is a devastating disease, not only for the victims, but their friends and family. Last year unpaid caregivers gave an estimated 17.4 billion hours towards caring for Alzheimer’s patients.
A point that is unfailingly raised in discussions about diagnosing Alzheimer’s disease is the fact that there is no cure. The few drugs that doctors prescribe are only meant to treat symptoms by improving cognitive function or decreasing anxiety. There’s a lot of people who don’t want to know if they’re at high risk for Alzheimer’s disease if they know it can’t be reversed. Fair enough. But whether or not you want to know right now, when there is a cure for Alzheimer’s, it’ll be nice to know we have tools to detect it and detect it early.
Image credits: alzheimer's association and modified from journal of the american medical association
images 1 and 2: JAMA
image 3: Alzheimer's
video: Alzheimer's by the numbers
Peter Murray was born in Boston in 1973. He earned a PhD in neuroscience at the University of Maryland, Baltimore studying gene expression in the neocortex. Following his dissertation work he spent three years as a post-doctoral fellow at the same university studying brain mechanisms of pain and motor control. He completed a collection of short stories in 2010 and has been writing for Singularity Hub since March 2011.
Related Articles

AI Can Now Design and Run Thousands of Experiments Without Human Hands. We Aren’t Ready for the Risk to Biosecurity.

Three Countries Own the Lithium Market. An MIT Startup Wants to Break Their Grip.

How Fast Are You Aging? New Genetic Clock May Have the Answer
What we’re reading