Human Mini-Brains Grafted Into Injured Rats Restored Their Sight
Share
Nearly a decade ago, mini-brains shot onto the neuroscience scene with a hefty promise: understanding the developing brain and restoring injured brains.
Known as brain organoids, these tiny clumps of brain tissue—roughly the size of a lentil—look nothing like the three-pound organ piloting our lives. Yet under the surface, they behave eerily similar to the brain of a human fetus. Their neurons spark with electrical activity. They readily integrate with—and subsequently control—muscles, at least in a dish. Similar to full-blown brains, they give birth to new neurons. Some even develop the six-layered structure of the human cortex—the wrinkly, outermost layer of the brain that supports thought, reasoning, judgment, speech, and perhaps even consciousness.
Yet a critical question haunts neuroscientists: can these Frankenstein bits of brain tissue actually restore an injured brain?
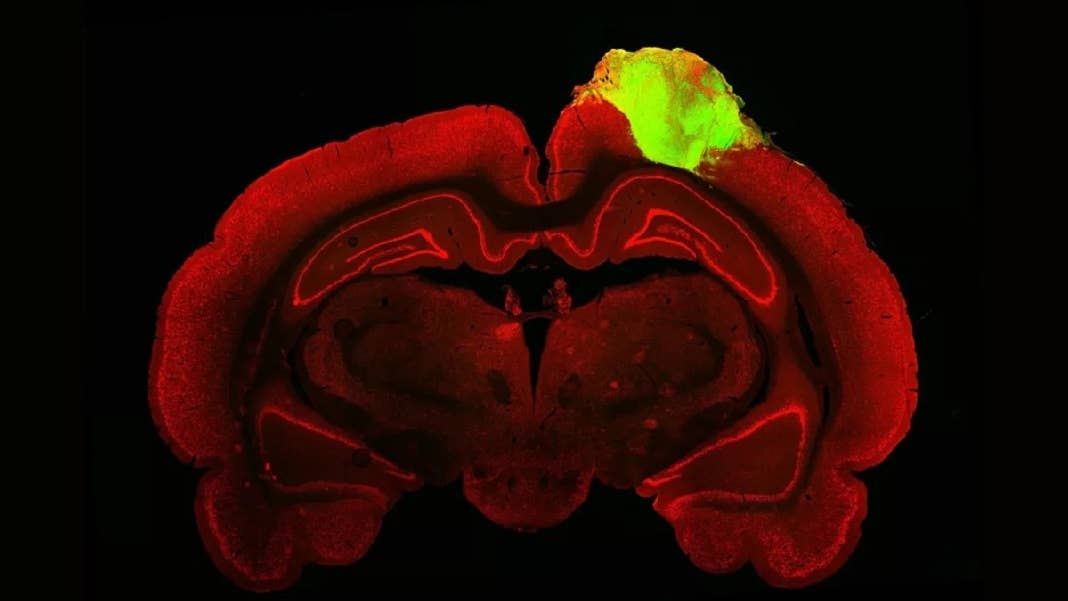
A study published in Cell Stem Cell this month concluded that they can. Using brain organoids made from human cells, a team led by Dr. Han-Chiao Isaac Chen at the University of Pennsylvania transplanted the mini-brains into adult rats with substantial damage to their visual cortex—the area that supports vision.
In just three months, the mini-brains merged with the rats’ brains. When the team shone flashing lights for the animals, the organoids spiked with electrical activity. In other words, the human mini-brain received signals from the rats’ eyes.
It’s not just random noise. Similar to our visual cortex, some of the mini-brain’s neurons gradually developed a preference for light shone at a particular orientation. Imagine looking at a black and white windmill blow toy as your eyes adjust to the different moving stripes. It sounds simple, but the ability of your eyes to adjust—dubbed “orientation selection”—is a sophisticated level of visual processing that’s critical to how we perceive the world.
The study is one of the first to show that mini-brain tissue can integrate with an injured adult host and perform its intended function. Compared to previous attempts at stem cell transplants, the artificial tissues could substitute an injured or degenerating piece of the brain in the future—but many caveats remain.
“Neural tissues have the potential to rebuild areas of the injured brain,” said Chen. “We haven't worked everything out, but this is a very solid first step.”
A Mini-Brain’s Mini-Life
Brain organoids have had a hell of a ride. First engineered in 2014, they immediately captured the interest of neuroscientists as an unprecedented model of the brain.
The quasi-brains are made from multiple sources to mimic different areas of the brain. One immediate use was to combine the technology with iPSCs (induced pluripotent stem cells) to study neurodevelopmental disorders, such as schizophrenia or autism.
Here, a patient’s skin cells are transformed back into a stem-cell-like state, which can be further grown into a 3D tissue of their brain. Because the person and the mini-brain share the same genes, it’s possible to partially duplicate the person’s brain during development—and potentially hunt down new cures.
Since their birth, mini-brains have now expanded in size, age, and sophistication. One major leap was a consistent blood supply. Our brains are intimately intertwined with blood vessels, feeding our neurons and neural networks with oxygen and nutrients to supply energy. The breakthrough came in 2017, when several teams showed that transplanting human organoids into rodent brains triggered the host’s blood vessels to integrate and “feed” the structured brain tissue, allowing it to further develop into the intricate brain architecture inside the host. The studies sparked a firestorm of discussion within the field, with bioethicists and researchers alike wondering if human organoids could change a rodent’s perception or behavior.
Chen had a different, if more challenging idea. Most previous studies transplanted mini-brains into infant rodents to nurture the organoids and ease their merger with the developing brain.
Adult brains, in contrast, are far more ridged. Highly intertwined neural circuits—including their signaling and functions—are already established. Even when injured, when the brain is ready for repair, shoving in extra bits of human organoid grafts like a Band-Aid could support broken neural circuits—or interfere with established ones.
Chen’s new study put the theory to the test.
An Unexpected Merger
To start, the team cultivated brain organoids with a renewable human stem cell line. Using a previously validated chemical recipe, the cells were coaxed into mini-brains that mimic the frontal parts of the cortex (around the forehead).
By day 80, the team saw rudimentary cortical layers in the organoid, along with cells organized in a way that resembled a developing brain. They then transplanted the organoids into the damaged visual cortex of young adult rats.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


Just one month after transplant, the host’s blood vessels merged with the human tissue, supplying it with much-needed oxygen and nutrients and allowing it to further grow and mature. The mini-brains developed a myriad of different brain cells—not just neurons, but also “supporting” brain cells such as astrocytes and specialized immune cells dubbed microglia. The latter two are far from dispensable: they have been implicated in brain aging, Alzheimer’s disease, inflammation, and cognition.
But can the transplanted human mini-brain function inside a rat?
In a first test, the team used a popular tracer to map the connections between the organoid and the animal’s eye. Similar to a dye, the tracer is a virus that hops between neural connections—dubbed synapses—while carrying a protein that glows a bright green under a fluorescent microscope. Like a highlighted route on Google Maps, the light stream clearly connected all the way to the transplanted mini-brain, meaning that its circuitry linked up, through multiple synapses, to the rats’ eyes.
Second question: could the transplanted tissue help the rat “see”? In six out of eight animals, turning the lights on or off triggered an electrical response, suggesting the human neurons responded to outside stimulation. The pattern of the electrical activity resembled natural ones seen in the visual cortex, “suggesting that organoid neurons have a comparable potential for light responsiveness to visual cortex neuron,” the authors said.
In another test, the grafts developed “picky” neurons that preferred a specific orientation selectivity for light—a quirk embedded inside our ability to perceive the world. When tested with different light gratings that flickered from black to white, the grafted neurons’ overall preference mimicked that of normal, healthy neurons.
“We saw that a good number of neurons within the organoid responded to specific orientations of light, which gives us evidence that these organoid neurons were able to not just integrate with the visual system, but they were able to adopt very specific functions of the visual cortex,” said Chen.
Plug-and-Play Brain Tissue?
The study shows that mini-brains can rapidly establish neural networks with the host’s brain, at a rate far faster than transplanting individual stem cells. It suggests a powerful use for the technology: repairing damaged brains at unprecedented speed.
Many questions remain. For one, the study was conducted in rats dosed with immunosuppressants to inhibit rejection. The hope for mini-brains is that they’ll be cultured from a patient’s own cells, eliminating the need for immunosuppressant drugs—a hope yet to be fully tested. Another problem is how to best match the mini-brain’s “age” to its host’s, so as not to disrupt the person’s intrinsic neural signals.
The team’s next step is to support other damaged brain regions using mini-brains, particularly damage due to degeneration from age or disease. Adding non-invasive technologies, such as neuromodulation or visual “rehabilitation” of the neurons, could further help the transplant integrate into the host’s circuit and potentially elevate their function.
“Now, we want to understand how organoids could be used in other areas of the cortex, not just the visual cortex, and we want to understand the rules that guide how organoid neurons integrate with the brain so that we can better control that process and make it happen faster,” said Chen.
Image Credit: Jgamadze et al.
Dr. Shelly Xuelai Fan is a neuroscientist-turned-science-writer. She's fascinated with research about the brain, AI, longevity, biotech, and especially their intersection. As a digital nomad, she enjoys exploring new cultures, local foods, and the great outdoors.
Related Articles

How Fast Are You Aging? New Genetic Clock May Have the Answer

Photosynthetic Drops Soothe Dry Eyes With Sunlight

A Revolutionary Cancer Treatment Could Transform Autoimmune Disease
What we’re reading