How to Make Sense of Uncertainty in a Coronavirus World

Share
As the internet churns with information about Covid-19, about the virus that causes the disease, and about what we’re supposed to do to fight it, it can be difficult to see the forest for the trees. What can we realistically expect for the rest of 2020? And how do we even know what’s realistic?
Today, humanity’s primary, ideal goal is to eliminate the virus, SARS-CoV-2, and Covid-19. Our second-choice goal is to control virus transmission. Either way, we have three big aims: to save lives, to return to public life, and to keep the economy functioning.
To hit our second-choice goal—and maybe even our primary goal—countries are pursuing five major public health strategies. Note that many of these advances cross-fertilize: for example, advances in virus testing and antibody testing will drive data-based prevention efforts.
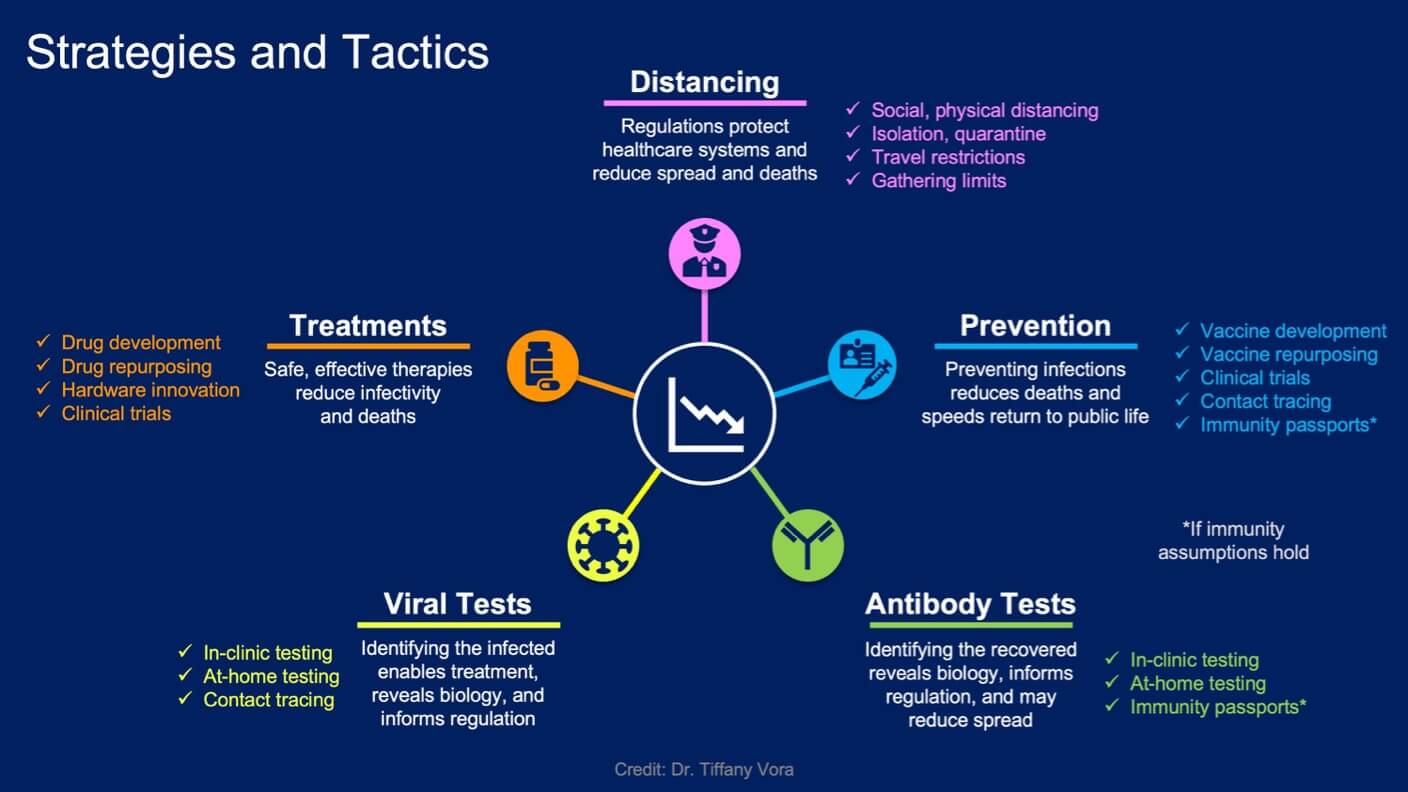
Five major public health strategies are underway to bring Covid-19 under control and to contain the spread of SARS-CoV-2.
These strategies arise from things we can control based on the things that we know at any given moment. But what about the things we can’t control and don’t yet know?
The biology of the virus and how it interacts with our bodies is what it is, so we should seek to understand it as thoroughly as possible. How long any immunity gained from prior infection lasts—and indeed whether people develop meaningful immunity at all after infection—are open questions urgently in need of greater clarity. Similarly, right now it’s important to focus on understanding rather than making assumptions about environmental factors like seasonality.
But the biggest question on everyone’s lips is, “When?” When will we see therapeutic progress against Covid-19? And when will life get “back to normal”? There are lots of models out there on the internet; which of those models are right? The simple answer is “none of them.” That’s right—it’s almost certain that every model you’ve seen is wrong in at least one detail, if not all of them. But modeling is meant to be a tool for deeper thinking, a way to run mental (and computational) experiments before—and while—taking action. As George E. P. Box famously wrote in 1976, “All models are wrong, but some are useful.”
Here, we’re seeking useful insights, as opposed to exact predictions, which is why we’re pulling back from quantitative details to get at the mindsets that will support agency and hope. To that end, I’ve been putting together timelines that I believe will yield useful expectations for the next year or two—and asking how optimistic I need to be in order to believe a particular timeline.
For a moderately optimistic scenario to be relevant, breakthroughs in science and technology come at paces expected based on previous efforts and assumptions that turn out to be basically correct; accessibility of those breakthroughs increases at a reasonable pace; regulation achieves its desired effects, without major surprises; and compliance with regulations is reasonably high.
In contrast, if I’m being highly optimistic, breakthroughs in science and technology and their accessibility come more quickly than they ever have before; regulation is evidence-based and successful in the first try or two; and compliance with those regulations is high and uniform. If I’m feeling not-so-optimistic, then I anticipate serious setbacks to breakthroughs and accessibility (with the overturning of many important assumptions), repeated failure of regulations to achieve their desired outcomes, and low compliance with those regulations.
The following scenarios outline the things that need to happen in the fight against Covid-19, when I expect to see them, and how confident I feel in those expectations. They focus on North America and Europe because there are data missing about China’s 2019 outbreak and other regions are still early in their outbreaks. Perhaps the most important thing to keep in mind throughout: We know more today than we did yesterday, but we still have much to learn. New knowledge derived from greater study and debate will almost certainly inspire ongoing course corrections.
As you dive into the scenarios below, practice these three mindset shifts. First, defeating Covid-19 will be a marathon, not a sprint. We shouldn’t expect life to look like 2019 for the next year or two—if ever. As Ed Yong wrote recently in The Atlantic, “There won’t be an obvious moment when everything is under control and regular life can safely resume.” Second, remember that you have important things to do for at least a year. And third, we are all in this together. There is no “us” and “them.” We must all be alert, responsive, generous, and strong throughout 2020 and 2021—and willing to throw away our assumptions when scientific evidence invalidates them.
The Middle Way: Moderate Optimism
Let’s start with the case in which I have the most confidence: moderate optimism.
This timeline considers milestones through late 2021, the earliest that I believe vaccines will become available. The “normal” timeline for developing a vaccine for diseases like seasonal flu is 18 months, which leads to my projection that we could potentially have vaccines as soon as 18 months from the first quarter of 2020. While Melinda Gates agrees with that projection, others (including AI) believe that 3 to 5 years is far more realistic, based on past vaccine development and the need to test safety and efficacy in humans. However, repurposing existing vaccines against other diseases—or piggybacking off clever synthetic platforms—could lead to vaccines being available sooner. I tried to balance these considerations for this moderately optimistic scenario. Either way, deploying vaccines at the end of 2021 is probably much later than you may have been led to believe by the hype engine. Again, if you take away only one message from this article, remember that the fight against Covid-19 is a marathon, not a sprint.
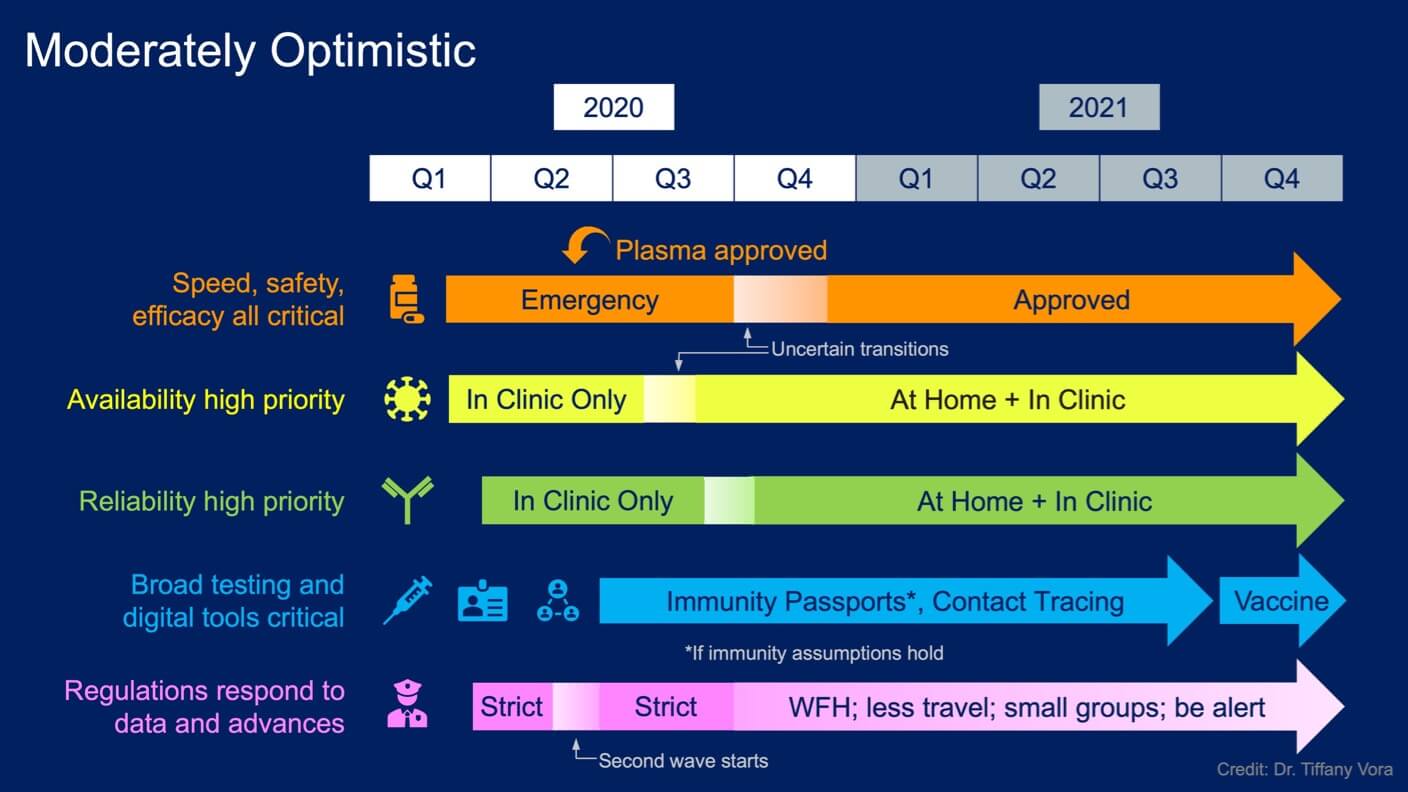
Here, I've visualized a moderately optimistic scenario as a baseline. Think of these timelines as living guides, as opposed to exact predictions. There are still many unknowns. More or less optimistic views (see below) and new information could shift these timelines forward or back and change the details of the strategies.
Based on current data, I expect that the first wave of Covid-19 cases (where we are now) will continue to subside in many areas, leading governments to ease restrictions in an effort to get people back to work. We’re already seeing movement in that direction, with a variety of benchmarks and changes at state and country levels around the world. But depending on the details of the changes, easing restrictions will probably cause a second wave of sickness (see Germany and Singapore), which should lead governments to reimpose at least some restrictions.
In tandem, therapeutic efforts will be transitioning from emergency treatments to treatments that have been approved based on safety and efficacy data in clinical trials. In a moderately optimistic scenario, assuming clinical trials currently underway yield at least a few positive results, this shift to mostly approved therapies could happen as early as the third or fourth quarter of this year and continue from there. One approval that should come rather quickly is for plasma therapies, in which the blood from people who have recovered from Covid-19 is used as a source of antibodies for people who are currently sick.
Companies around the world are working on both viral and antibody testing, focusing on speed, accuracy, reliability, and wide accessibility. While these tests are currently being run in hospitals and research laboratories, at-home testing is a critical component of the mass testing we’ll need to keep viral spread in check. These are needed to minimize the impact of asymptomatic cases, test the assumption that infection yields resistance to subsequent infection (and whether it lasts), and construct potential immunity passports if this assumption holds. Testing is also needed for contact tracing efforts to prevent further spread and get people back to public life. Finally, it’s crucial to our fundamental understanding of the biology of SARS-CoV-2 and Covid-19.
We need tests that are very reliable, both in the clinic and at home. So, don’t go buying any at-home test kits just yet, even if you find them online. Wait for reliable test kits and deeper understanding of how a test result translates to everyday realities. If we’re moderately optimistic, in-clinic testing will rapidly expand this quarter and/or next, with the possibility of broadly available, high-quality at-home sampling (and perhaps even analysis) thereafter.
Note that testing is not likely to be a “one-and-done” endeavor, as a person’s infection and immunity status change over time. Expect to be testing yourself—and your family—often as we move later into 2020.
Testing data are also going to inform distancing requirements at the country and local levels. In this scenario, restrictions—at some level of stringency—could persist at least through the end of 2020, as most countries are way behind the curve on testing (Iceland is an informative exception). Governments will likely continue to ask citizens to work from home if at all possible; to wear masks or face coverings in public; to employ heightened hygiene and social distancing in workplaces; and to restrict travel and social gatherings. So while it’s likely we’ll be eating in local restaurants again in 2020 in this scenario, at least for a little while, it’s not likely we’ll be heading to big concerts any time soon.
The Extremes: High and Low Optimism
How would high and low levels of optimism change our moderately optimistic timeline? The milestones are the same, but the time required to achieve them is shorter or longer, respectively. Quantifying these shifts is less important than acknowledging and incorporating a range of possibilities into our view. It pays to pay attention to our bias. Here are a few examples of reasonable possibilities that could shift the moderately optimistic timeline.
When vaccines become available
Vaccine repurposing could shorten the time for vaccines to become available; today, many vaccine candidates are in various stages of testing. On the other hand, difficulties in manufacture and distribution, or faster-than-expected mutation of SARS-CoV-2, could slow vaccine development. Given what we know now, I am not strongly concerned about either of these possibilities—drug companies are rapidly expanding their capabilities, and viral mutation isn’t an urgent concern at this time based on sequencing data—but they could happen.
At first, governments will likely supply vaccines to essential workers such as healthcare workers, but it is essential that vaccines become widely available around the world as quickly and as safely as possible. Overall, I suggest a dose of skepticism when reading highly optimistic claims about a vaccine (or multiple vaccines) being available in 2020. Remember, a vaccine is a knockout punch, not a first line of defense for an outbreak.
Be Part of the Future
Sign up to receive top stories about groundbreaking technologies and visionary thinkers from SingularityHub.


When testing hits its stride
While I am confident that testing is a critical component of our response to Covid-19, reliability is incredibly important to testing for SARS-CoV-2 and for immunity to the disease, particularly at home. For an individual, a false negative (being told you don’t have antibodies when you really do) could be just as bad as a false positive (being told you do have antibodies when you really don’t). Those errors are compounded when governments are trying to make evidence-based policies for social and physical distancing.
If you’re highly optimistic, high-quality testing will ramp up quickly as companies and scientists innovate rapidly by cleverly combining multiple test modalities, digital signals, and cutting-edge tech like CRISPR. Pop-up testing labs could also take some pressure off hospitals and clinics.
If things don’t go well, reliability issues could hinder testing, manufacturing bottlenecks could limit availability, and both could hamstring efforts to control spread and ease restrictions. And if it turns out that immunity to Covid-19 isn’t working the way we assumed, then we must revisit our assumptions about our path(s) back to public life, as well as our vaccine-development strategies.
How quickly safe and effective treatments appear
Drug development is known to be long, costly, and fraught with failure. It’s not uncommon to see hope in a drug spike early only to be dashed later on down the road. With that in mind, the number of treatments currently under investigation is astonishing, as is the speed through which they’re proceeding through testing. Breakthroughs in a therapeutic area—for example in treating the seriously ill or in reducing viral spread after an infection takes hold—could motivate changes in the focus of distancing regulations.
While speed will save lives, we cannot overlook the importance of knowing a treatment’s efficacy (does it work against Covid-19?) and safety (does it make you sick in a different, or worse, way?). Repurposing drugs that have already been tested for other diseases is speeding innovation here, as is artificial intelligence.
Remarkable collaborations among governments and companies, large and small, are driving innovation in therapeutics and devices such as ventilators for treating the sick.
Whether government policies are effective and responsive
Those of us who have experienced lockdown are eager for it to be over. Businesses, economists, and governments are also eager to relieve the terrible pressure that is being exerted on the global economy. However, lifting restrictions will almost certainly lead to a resurgence in sickness.
Here, the future is hard to model because there are many, many factors at play, and at play differently in different places—including the extent to which individuals actually comply with regulations.
Reliable testing—both in the clinic and at home—is crucial to designing and implementing restrictions, monitoring their effectiveness, and updating them; delays in reliable testing could seriously hamper this design cycle. Lack of trust in governments and/or companies could also suppress uptake. That said, systems are already in place for contact tracing in East Asia. Other governments could learn important lessons, but must also earn—and keep—their citizens’ trust.
Expect to see restrictions descend and then lift in response to changes in the number of Covid-19 cases and in the effectiveness of our prevention strategies. Also expect country-specific and perhaps even area-specific responses that differ from each other. The benefit of this approach? Governments around the world are running perhaps hundreds of real-time experiments and design cycles in balancing health and the economy, and we can learn from the results.
A Way Out
As Jeremy Farrar, head of the Wellcome Trust, told Science magazine, “Science is the exit strategy.” Some of our greatest technological assistance is coming from artificial intelligence, digital tools for collaboration, and advances in biotechnology.
Our exit strategy also needs to include empathy and future visioning—because in the midst of this crisis, we are breaking ground for a new, post-Covid future.
What do we want that future to look like? How will the hard choices we make now about data ethics impact the future of surveillance? Will we continue to embrace inclusiveness and mass collaboration? Perhaps most importantly, will we lay the foundation for successfully confronting future challenges? Whether we’re thinking about the next pandemic (and there will be others) or the cascade of catastrophes that climate change is bringing ever closer—it’s important to remember that we all have the power to become agents of that change.
Special thanks to Ola Kowalewski and Jason Dorrier for significant conversations.
Image Credit: Drew Beamer / Unsplash
Dr. Tiffany Vora is an educator, writer, research scientist, and entrepreneur who is excited to bring her diversity of experience to Singularity University as faculty and vice chair of Medicine and Digital Biology. Tiffany is passionate about translating the leading edge of science and medicine into actionable insights for individuals, companies, and society. After earning undergraduate degrees in Biology and Chemistry at New York University, Tiffany worked on cutting-edge drug-discovery technologies at Bristol-Myers Squibb. Her PhD research in the Department of Molecular Biology at Princeton University, which was funded through NASA, brought her into the emerging fields of genomics, systems biology, and computational biology. With the Mars Society, Tiffany designed and supported biology- and health-related research in Mars simulations in extreme environments. Her interest in the cultural and ethical shifts that accompany new technologies and new ways of thinking then led her to the American University of Cairo as a visiting assistant professor, where she spearheaded curriculum development for core classes in scientific thinking as well as computational biology classes for non-programmers. Upon her return to the United States, Tiffany founded Bayana Science, an editing, writing, and consulting company dedicated to excellence in science communication. Tiffany also served as an instructor for the Department of Bioengineering at Stanford University. She has contributed to literally thousands of grant proposals, research articles, presentations, textbooks, and other works spanning medicine, computer science, applied physics, chemistry, nanotechnology, and the life sciences. Tiffany also co-founded Three Body Labs, which is dedicated to fostering thought leadership with a global impact. At Singularity University, Tiffany previously served as full-time faculty as well as the faculty director of Singularity’s global ecosystem of experts and thought leaders. Through Singularity, Tiffany has mentored startup founders, inspired companies to tackle humanity’s grand challenges, and supported governments and other organizations to prepare for a radically different future of life on Earth (and elsewhere). She has particularly focused on nurturing, amplifying, and connecting women and people of color around the world—because diversity drives creativity and innovation. Tiffany’s expertise in biology and medicine encompasses fields as diverse as CRISPR, the microbiome, genetics, science fiction, climate change, longevity, human space biology, agriculture, AR/VR in the life sciences, ancient molecules, biophysics, environmental monitoring, tissue engineering, biohacking, and the quantitative analysis of large biological datasets. Tiffany loves encountering the natural world through hiking and scuba diving. She travels extensively with her family, seeking out new experiences and cultures. She enjoys sharing her passions through teaching, writing, and public speaking.
Related Articles

US Issues Grand Challenge: The First Fault-Tolerant Quantum Computer by 2028
Five Ways Quantum Technology Could Shape Everyday Life

Chatbots ‘Optimized to Please’ Make Us Less Likely to Admit When We’re Wrong
What we’re reading